Understanding the electronic configuration of elements is fundamental to grasping their chemical properties and behaviors. Among the elements, oxygen (O) holds a special place due to its ubiquitous presence in the Earth's atmosphere and its crucial role in biological processes. This blog post delves into the electronic configuration of oxygen, its significance, and how it influences the element's reactivity and bonding capabilities.
What is Electronic Configuration?
Electronic configuration refers to the arrangement of electrons in the orbitals of an atom. This arrangement is governed by the principles of quantum mechanics and is essential for understanding the chemical behavior of elements. The electronic configuration is typically written using a notation that specifies the energy levels (shells) and sublevels (orbitals) where the electrons are located.
The Electronic Configuration of Oxygen
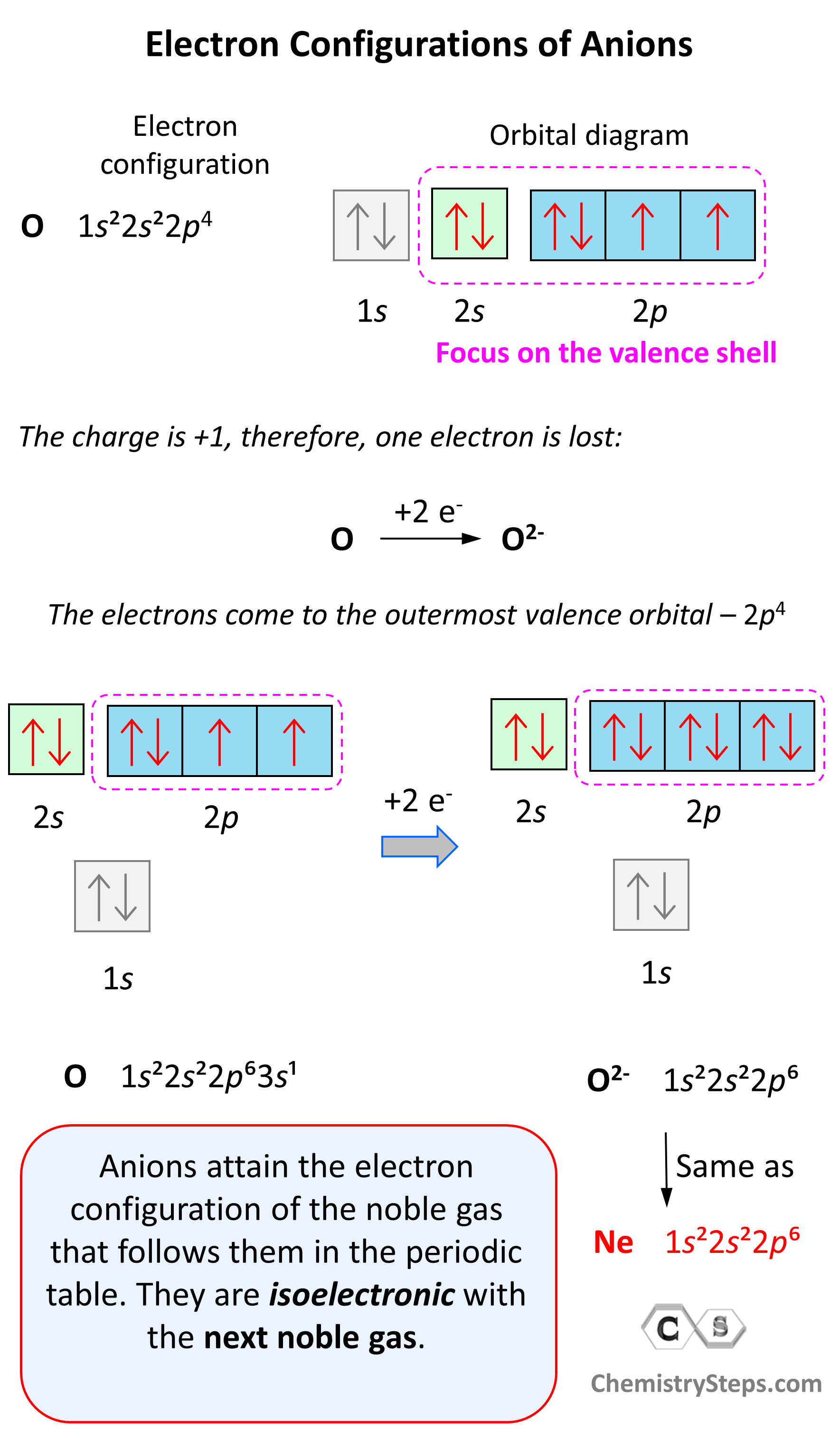
The electronic configuration of oxygen can be determined by following the Aufbau principle, which states that electrons fill the lowest energy levels first before moving to higher energy levels. Oxygen has an atomic number of 8, meaning it has 8 protons and 8 electrons. The electronic configuration of oxygen is:
1s2 2s2 2p4
Breaking this down:
- The 1s orbital is the lowest energy level and can hold up to 2 electrons.
- The 2s orbital is the next lowest energy level and also holds up to 2 electrons.
- The 2p orbital can hold up to 6 electrons, but in oxygen, it holds 4 electrons.
This configuration indicates that oxygen has 2 electrons in the 1s orbital, 2 electrons in the 2s orbital, and 4 electrons in the 2p orbital.
Significance of the Electronic Configuration of Oxygen
The electronic configuration of oxygen plays a critical role in determining its chemical properties. Oxygen’s outer shell (valence shell) contains 6 electrons, which means it needs 2 more electrons to achieve a stable octet configuration. This makes oxygen highly reactive and prone to forming bonds with other elements to complete its outer shell.
Bonding and Reactivity
Oxygen’s electronic configuration influences its bonding and reactivity in several ways:
- Covalent Bonding: Oxygen can form covalent bonds with other non-metals. For example, in water (H2O), oxygen forms covalent bonds with two hydrogen atoms. Each hydrogen atom contributes one electron to the bond, allowing oxygen to achieve a stable octet configuration.
- Ionic Bonding: Oxygen can also form ionic bonds with metals. For instance, in sodium oxide (Na2O), oxygen forms ionic bonds with sodium atoms. Each sodium atom donates one electron to oxygen, resulting in a stable configuration for both elements.
- Oxidation States: Oxygen can exhibit different oxidation states, the most common being -2. This is because oxygen typically gains 2 electrons to complete its octet, resulting in a -2 charge.
Oxygen in Biological Systems
Oxygen’s electronic configuration is crucial for its role in biological systems. In the human body, oxygen is transported by hemoglobin in red blood cells. Hemoglobin contains iron, which binds to oxygen molecules, forming a stable complex. This process is essential for respiration, where oxygen is used to produce energy in the form of ATP.
Oxygen in the Environment
Oxygen’s electronic configuration also influences its behavior in the environment. In the Earth’s atmosphere, oxygen exists primarily as diatomic molecules (O2). This form is stable due to the covalent double bond between the two oxygen atoms. However, oxygen can also exist in other forms, such as ozone (O3), which plays a crucial role in protecting the Earth from harmful ultraviolet radiation.
Applications of Oxygen
Oxygen’s unique electronic configuration makes it valuable in various applications:
- Medical Use: Oxygen is used in medical settings to treat patients with respiratory issues. It is administered through masks or nasal cannulas to ensure adequate oxygen supply to the body.
- Industrial Use: Oxygen is used in industrial processes such as steelmaking, where it is used to remove impurities from molten metal. It is also used in welding and cutting processes.
- Environmental Use: Oxygen is essential for the combustion of fuels, making it a key component in energy production. It is also used in wastewater treatment to support the growth of aerobic bacteria that break down organic matter.
Comparing Oxygen with Other Elements
To better understand the electronic configuration of oxygen, it is helpful to compare it with other elements in the same period (row) of the periodic table. The following table shows the electronic configurations of elements in the second period:
| Element | Atomic Number | Electronic Configuration |
|---|---|---|
| Lithium (Li) | 3 | 1s2 2s1 |
| Beryllium (Be) | 4 | 1s2 2s2 |
| Boron (B) | 5 | 1s2 2s2 2p1 |
| Carbon © | 6 | 1s2 2s2 2p2 |
| Nitrogen (N) | 7 | 1s2 2s2 2p3 |
| Oxygen (O) | 8 | 1s2 2s2 2p4 |
| Fluorine (F) | 9 | 1s2 2s2 2p5 |
| Neon (Ne) | 10 | 1s2 2s2 2p6 |
This comparison highlights how the electronic configuration changes as the number of electrons increases. Oxygen’s configuration places it between nitrogen and fluorine, with a unique set of properties that make it essential for life and various industrial processes.
🔍 Note: The electronic configuration of an element is a fundamental concept in chemistry that helps explain its chemical behavior and reactivity. Understanding the electronic configuration of oxygen provides insights into its role in biological systems, environmental processes, and industrial applications.
Oxygen’s electronic configuration is a cornerstone of its chemical behavior and reactivity. Its ability to form stable bonds with other elements makes it indispensable in various fields, from medicine to industry. By understanding the electronic configuration of oxygen, we gain a deeper appreciation for its role in sustaining life and driving technological advancements.
Related Terms:
- electronic configuration of periodic table
- complete electron configuration for oxygen
- electron configuration and periodic table
- electron configuration diagram for o2
- electron configurations chart
- full electron configuration for oxygen